|
And so ah, PK of eight is greater than the ph of 7.4. Notice that our amino group eyes right here the n terminus, and it has a PKF eight. Hold on, let me get rid of this color here. And so what we need to do is check our amino group on here and notice that our end terminus are amino group. And so we're gonna be comparing the Pete, the PKK's of each of these ionized able groups to the pH that's given to us of 74 So let's first start with the amino group on the far left over here. So basically, we have 12345 ionized able groups that we need to consider for this peptide. But it's on the end and we know that it's gonna have a nine invisible car boxful group. Well, and then Glutamine does not have a nine Izabal our group. So is history into our group that we need to consider So I'm leaving these blank spaces here so that you know that these air, the ionized able groups that we need to consider ah Spartak Acid remembers a negatively charged, acidic amino acid. So we know that it's our group we also need to consider to see if it's gonna be ionized or not. So we know Argentine over here is gonna have a, uh, amino group that we need to consider that's ionized able and Argentine, we know is one of the positively charged basic amino acids. So let's go ahead and rewrite the peptide over here just so that we have a little bit more space and so we need to just consider it's ionized able groups. We just need to consider all of its ionized herbal groups. Alright, So that being said, we're gonna estimate the net charge of this peptide, and we don't need to redraw the peptide. And so again, if you're using the wrong set of PK is, then that could affect your calculation on the estimation of the charges, so just make sure that you're using the right set of PKS and we're gonna be using this table here for all of our, um, P K s for amino acid residues. But for amino acid residues noticed that the PK is 3.5, which is shifted 1.5 units and again, that can affect the charges when we go to try to determine them. And so I remember that we said that normally the car boxful group has a PK of about two in the ballpark of about two. 
And this is specifically for, um, you know, acid residues. And then over here in this blue chart over here, what we have is the PK a values. And in this example, it says toe estimate the net charge of the peptide at physiological pH, which is 7.4 and the peptide years Argentine history, a Spartak acid and glutamine. Alright, so that being said, let's go to our example. Then you need thio zone in a little bit more and make sure that you're using the right set of PKS. K s for amino acid residues than for free amino acids. But if you're a professor expects you to use an entirely different set of P. You're good to go, and you don't have to worry about anything. And so if you're professor uses the same set of PKK's for free amino acids and amino acid residues, that's totally fine. So you wanna make sure that you're using the P K A values for amino acid residues and not for free amino acids. And so if that confused you a little bit, all I really want you guys to know is that I want you to double check and make sure that you're using the correct set of PK A values. And so when we go to compare the P K s to the P H, if the P K s are shifted by several units, that's going to affect the net charge. It can affect the polarity of the atoms, and it can actually shift the PK a values of amino acid residues by several units. So it's the immediate area surrounding an atom or molecule, and the micro environment can actually shift. And so all the micro environment is is it's the immediate vicinity surrounding an atom or molecule. 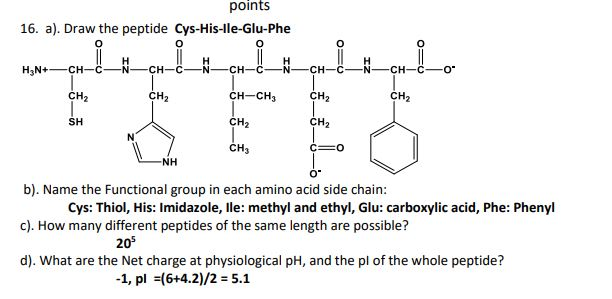
And the reason is because there is a unique micro environment for every single peptide. You can Onley estimated unless you determine it experimentally. And so the last thing I want to leave you guys off with before we get to our example is the fact this idea that the net charge of a known poly peptide can Onley be estimated. And the internal amino acid residues lack Alfa amino or Alfa car boxful, ionized able groups. Just like we did in some of our previous lessons and then also recall that on Lee, the very first and the very last amino acid residues in the chain have free Orion, Izabal, Alfa Amino or Alfa car boxes groups, respectively. And so recall that the way that we determine the ionization oven Ionized Herbal Group is just by comparing the p k a of the ionized well group to the ph of the solution. So the net charge of a protein is actually dictated by the net charges of all of its ionized herbal groups. In this video, we're going to talk about how to determine the net charge of a peptide or protein.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
